Describe Isotopes Using the Atomic Model
There is an easy way to represent isotopes using the atomic symbols. Using the sim examine Natures mix of isotopes.
Isotope Basics Nidc National Isotope Development Center
The average atomic mass is the weighted average of all the isotopes of an element.
. The Bohr Model of the atom was a slightly more intuitive model that human language used to describe however it was also inaccurate. Isotopes and Atomic Mass Worksheet Answer Key. Describe a method to calculate the average atomic mass of the sample in the previous question using only the atomic masses of lithium-6 and lithium-7 without using the simulation.
This is because atoms of an element can differ in the number of neutrons Q. SP6 They will be using the model to describe how carbon dating works. All matter is made of atoms.
Up to 24 cash back In order to find the percent abundance given two isotopes and the atomic mass of an element you need to set the percent abundance of an isotope in a sample of 100 atoms equal to x. Masses of the isotopes and the quantity of each isotope. Write a note on isotopes of Uranium.
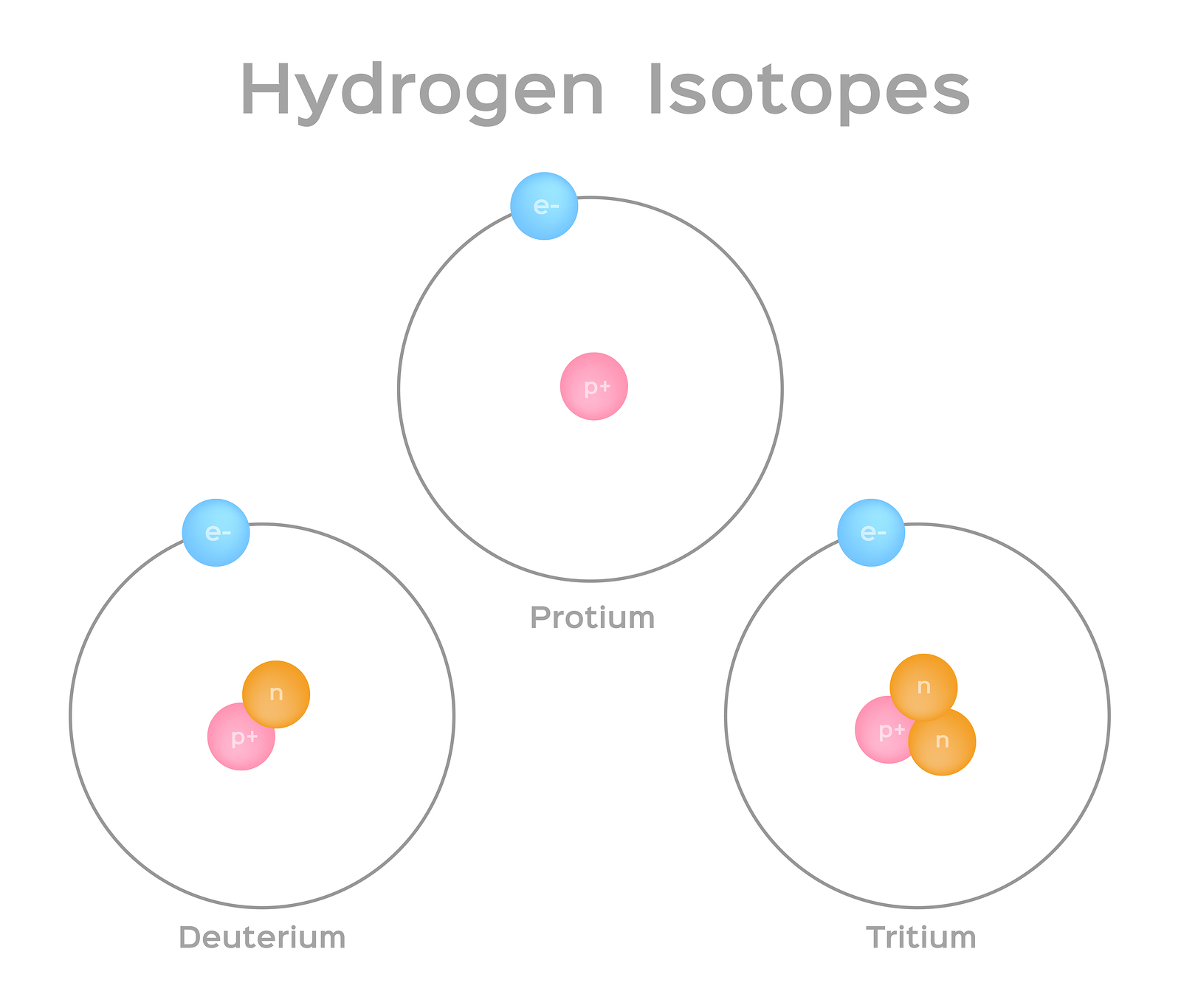
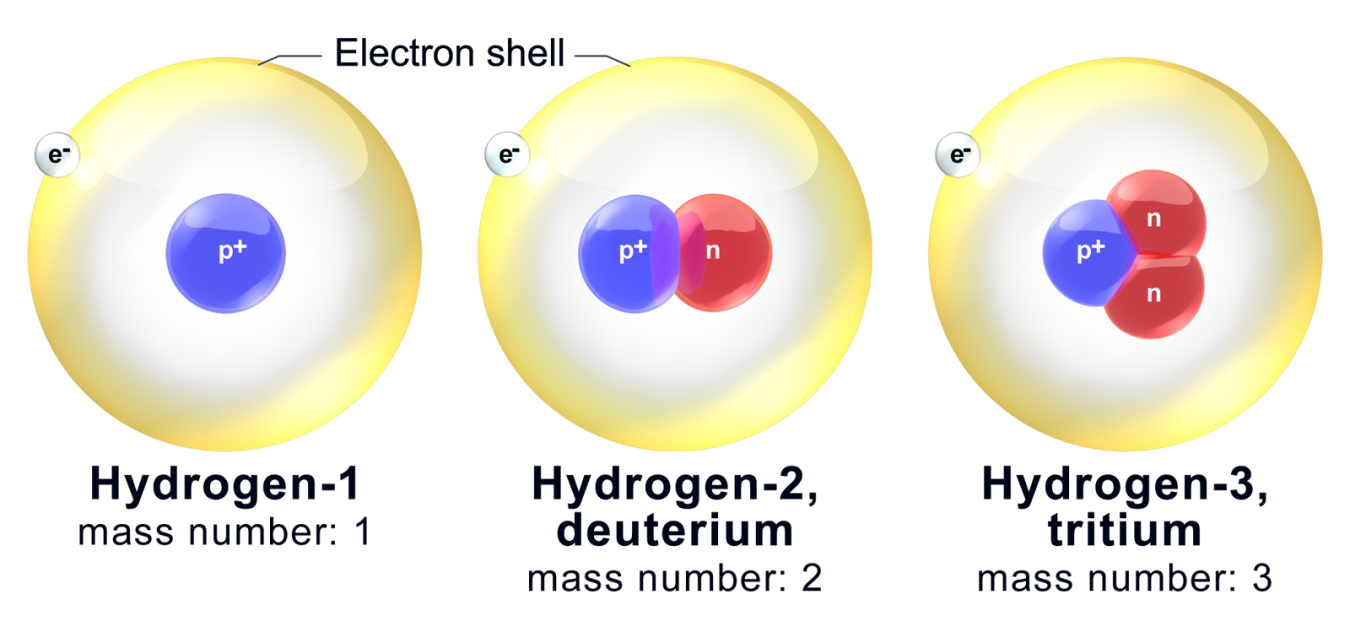
Isotopes are atoms of an element whose nuclei have the same atomic number but different mass number. Isotopes are atoms of the same element that have different numbers of neutrons but the same number of protons and electrons. Test your method by creating a few sample mixtures of isotopes with the sim and see if your method correctly predicts the average atomic mass of that sample from.
235 92 U or U-235. Use the table below to track your progress. Element Mass of 1 atom Average mass of 2 atoms sim Average mass of 3 atoms sim Atomic mass periodic table Beryllium Be 901218 amu 901218mu 901218 901218 Fluorine F 1899840 amu 1899840Amu 1899840 1899840 8.
63 cu has a mass of 62 929 599 amu and occurs 69 17 of the time 65 cu has a mass of 64 927 793 amu with a natural abundance. A mass number Zatomic number Use notation for writing isotope symbols. Atoms of one element are all the same.
In this activity students will define what isotopes are illustrate important characteristics and provide both examples and non-examples. Element Atomic mass and quantity of each isotope Average atomic mass of sample calculate yourself Average atomic mass of sample from simulation MODEL 3. Lithium has two naturally occurring isotopes.
Alkanes are named with the root based on the number of carbon. Modern atomic theory explains details about isotopes having a different number of neutrons and the same number of protons. Larger alkanes are named according to the Greek prefixes.
Atomic Mass of each isotope and abundance of each isotope 3. We use the construction. View Isotopes_Atomic-Mass_Guided-Inquiry_1 from SCIENCE 123456789 at Woodridge High School.
Natures mix of isotopes 1. Alkanes are named with the root based on the number of hydrogen atoms followed by the suffix -ane. Thus the concept of isotopes in which an element has different masses was a violation of the original idea.
12C 14 13C C a. Up to 24 cash back SP2 will be used because they are using a simulation to understand isotopes and stability. A Frayer Model is a perfect tool to deepen the understanding of isotopes.
The subatomic particles in the Bohr model of the atom are all. Thus for the isotope of carbon that has 6 protons and 6 neutrons the symbol is. Meth- eth- prop- and but- are the root names for alkanes with one two three and four carbons respectively.
ISOTOPES AND ATOMIC MASS MODEL 1. Using these data calculate the approximate atomic mass of lead. Because the identity of an atom is defined by its atomic.
Atoms cannot be broken down into smaller parts. It is defined as. Lithium-6 has an atomic mass of 6015 amu.
The stable isotopes have nuclei that do not decay to other isotopes on geologic timescales but may themselves be produced by. Which parts of Daltons theory were later found to be erroneous. Find step-by-step Chemistry solutions and your answer to the following textbook question.
Then multiply the mass number of each isotope by the abundance 100-x and x and set the final expression equal to the given. Protons Electrons Neutrons Atomic Number Atomic Mass Uranium-235. For example the relative atomic mass of chlorine is 355 rather than a whole number.
Lets Practice w isotopes. Use the periodic table. Isotopes are atoms of an element that differ in their number of neutrons.
Identify isotope using mass number and atomic number and relate to number of protons neutrons and electrons. The protons and neutrons are found in. The four isotopes of lead are shown below each with its percent by mass abundance and the composition of its nucleus.
Describe the changes in the atomic model based on Daltons atomic theory using your own words. Use the periodic table. Where X is the symbol of the element A is the mass number and Z is the atomic number.
1 1 2 Atomic Structure Describe protons neutrons and electrons from Isotope. Use the sim and the periodic table to complete the following table. Up to 24 cash back Characterize protons neutrons electrons by location relative charge relative mass p1 n1 e12000.
Daltons atomic theory is a set of postulates proposed to describe the structure and properties of. Isotopes is a Greek word isos means same and tope means place. Atoms contain three sub-atomic particles called protons neutrons.
John Daltons Atomic Theory 1804. Therefore the other is equal to 100-x. First used by Soddy.
SP5 will be used since they are calculating average atomic masses from mixtures of molecules. Beryllium Be and Fluorine F have only one stable isotope. Lets Practice with Ions.
To account for the existence of isotopes the second postulate of his atomic theory was modified to state that atoms of the same element must have identical chemical properties. This is because chlorine contains two different isotopes chlorine-35 and chlorine-37. The nuclear model The structure of the atom.
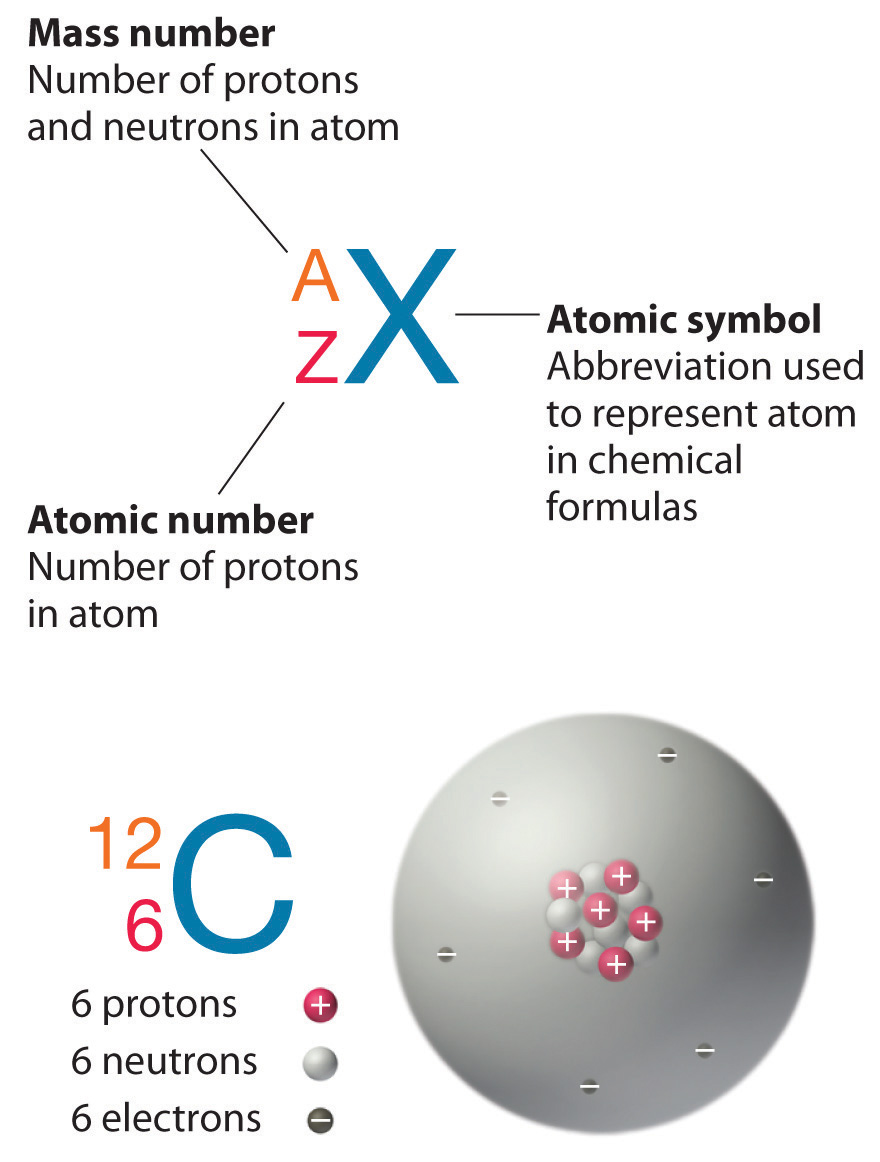

Comments
Post a Comment